February 2017 Sees Decline in Federal Personal Injury/Product Liability Suits
The latest available data from the federal courts show that during February 2017 the government reported 1,972 new personal injury filings for product liability. According to the case-by-case information analyzed by the Transactional Records Access Clearinghouse (TRAC), this number is down 28.1% over the previous month when the number of civil filings of this type totaled 2,744.
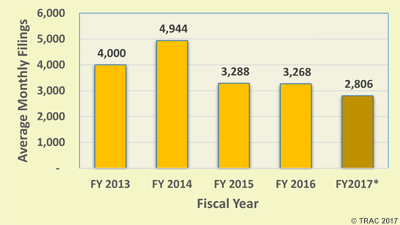
Figure 1. Average Monthly U.S. District Court Filings of
Personal Injury Product Liability Suits
(* five months only)
The comparisons of the number of civil filings are based on case-by-case court records which were compiled and analyzed by TRAC.
While February's filings were unusually low, the average monthly filings so far during FY 2017 were 2,806 - down from the average filings of 3,268 during all of FY 2016 and down from 4,944 during FY 2014. See Figure 1.
The month-by-month trend in civil filings for these matters going back five years is shown more clearly in Figure 2. The vertical bars in Figure 2 represent the number of civil filings of this type recorded each month. The superimposed line on the bars plots the six-month moving average so that natural fluctuations are smoothed out. Civil filings for February 2017 are down 30.1 percent from levels reported five years ago based upon the moving averages.
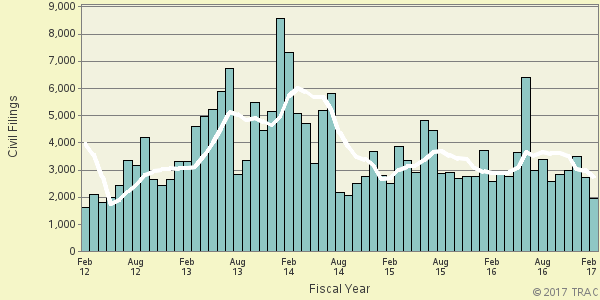
Figure 2. Civil Filings Over the Last Five Years
Specific Nature of Suits
The largest volume of personal injury/product liability suits filed during the first five months of FY 2017 involved health care and pharmaceuticals. During February 2017, nearly two out of every three suits (64%) were of this type. The largest number (531) were filed in the Eastern District of Louisiana where U.S District Judge Eldon E. Fallon is handling multidistrict litigation (MDL) involving the safety of Xarelto, a blood thinner developed by Johnson & Johnson's unit Janssen Pharmaceuticals and by Bayer.
The Southern District of West Virginia had the second largest number of suits (240) filed during February. Most of these suits are in a series of MDL cases involving the safety of transvaginal surgical mesh devices used to treat pelvic organ prolapse and stress urinary incontinence. These pelvic mesh MDL cases are being handled by U.S. District Judge Joseph Goodwin and involve suits against C.R. Bard, Inc. (MDL No. 2187), American Medical Systems, Inc. (MDL No. 2325), Boston Scientific Corp. (MDL No. 2326), Ethicon, Inc. (MDL No. 2327), Coloplast Corp. (MDL No. 2387), Cook Medical, Inc. (MDL No. 2440), and Neomedic (MDL 2511).
Table 1 compares the volume of personal injury/product liability suits over the past five years by the nature of these suits. While the overall volume of this litigation has declined, the health care and pharmaceuticals sector has seen a rise in volume of cases filed. During FY 2016, filings reached 20,287 suits, up from 7,457 filed during FY 2013. Already, during the first five months of FY 2017, filings of these types of cases have surpassed the total number that were brought during all of FY 2013.
| Personal Injury Product Liability Torts | FY2013 | FY 2014 | FY 2015 | FY 2016 | FY 2017* |
| Total | 47,999 | 59,333 | 39,457 | 39,212 | 14,031 |
| Airplane Product Liability | 28 | 53 | 48 | 90 | 10 |
| Asbestos Personal Injury Product Liability | 630 | 446 | 233 | 194 | 78 |
| Health Care / Pharmaceutical Personal Injury Product Liability | 7,457 | 13,172 | 15,305 | 20,287 | 7,000 |
| Marine Product Liability | 31 | 11 | 9 | 48 | 7 |
| Motor Vehicle Product Liability | 238 | 298 | 405 | 416 | 151 |
| Other Personal Injury- Product Liability | 39,615 | 45,353 | 23,457 | 18,177 | 6,785 |
Each month, TRAC offers a free report focused on one area of civil litigation in the U.S. district courts. In addition, subscribers to the TRACFed data service can generate custom reports by district, office, nature of suit or federal jurisdiction via the TRAC Data Interpreter.